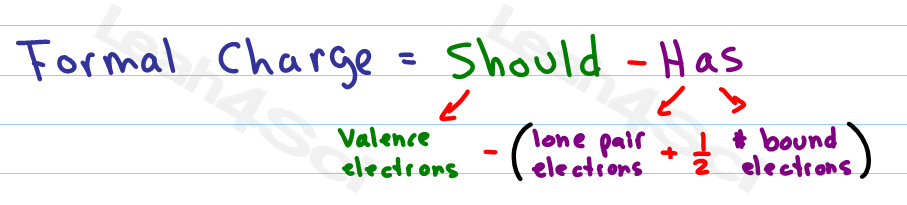
The C=O double bond is shorter than the C-O single bond. 
Higher bond orders generally correlate with shorter bond distances. Bond length is the equilibrium distance between two nuclei. This means that a C=O double bond is stronger than a C-O single bond, and the C=O double bond requires more energy to break than a C-O single bond. 
The stronger the bond, the more energy that is required to break the bond. Bond strength is a measured quantity: the energy (in kJ/mol) required to break a chemical bond, tabulated in Appendix G. The carbon is singly bonded to each hydrogen atom, which would give each CH bond orders of one.īond order is an index of bond strength: the higher the bond order, the stronger the bond. Again, experiments show that all three C–O bonds are exactly the same.Looking at the structure of formaldehyde we can see that there is a double bond between the central carbon atom and the oxygen atom giving a CO bond order of two. Because we can write three identical resonance structures, we know that the actual arrangement of electrons in the carbonate ion is the average of the three structures. This gives rise to three resonance forms of the carbonate ion. All oxygen atoms, however, are equivalent, and the double bond could form from any one of the three atoms. One oxygen atom must have a double bond to carbon to complete the octet on the central atom. Experiments show, however, that both N–O bonds in \(,\) provides a second example of resonance: A double bond between two atoms is shorter (and stronger) than a single bond between the same two atoms. 
If nitrite ions do indeed contain a single and a double bond, we would expect for the two bond lengths to be different. The electrons involved in the N–O double bond, however, are in different positions: You may have noticed that the nitrite anion in can have two possible structures with the atoms in the same positions. Also, it places the least electronegative atom in the center, and the negative charge on the more electronegative element (Guideline 4). However, the first arrangement of atoms is preferred because it has the lowest number of atoms with nonzero formal charges (Guideline 2). Note that the sum of the formal charges in each case is equal to the charge of the ion (–1). Possible Lewis structures and the formal charges for each of the three possible structures for the thiocyanate ion are shown here: The formal charges present in each of these molecular structures can help us pick the most likely arrangement of atoms. We can draw three possibilities for the structure: carbon in the center and double bonds, carbon in the center with a single and triple bond, and oxygen in the center with double bonds:Ĭomparing the three formal charges, we can definitively identify the structure on the left as preferable because it has only formal charges of zero (Guideline 1).Īs another example, the thiocyanate ion, an ion formed from a carbon atom, a nitrogen atom, and a sulfur atom, could have three different molecular structures: CNS –, NCS –, or CSN –. We know from our previous discussion that the less electronegative atom typically occupies the central position, but formal charges allow us to understand why this occurs. To see how these guidelines apply, let us consider some possible structures for carbon dioxide, CO 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
